Why Pick-Two?
Pick-Two Consultancy can…..
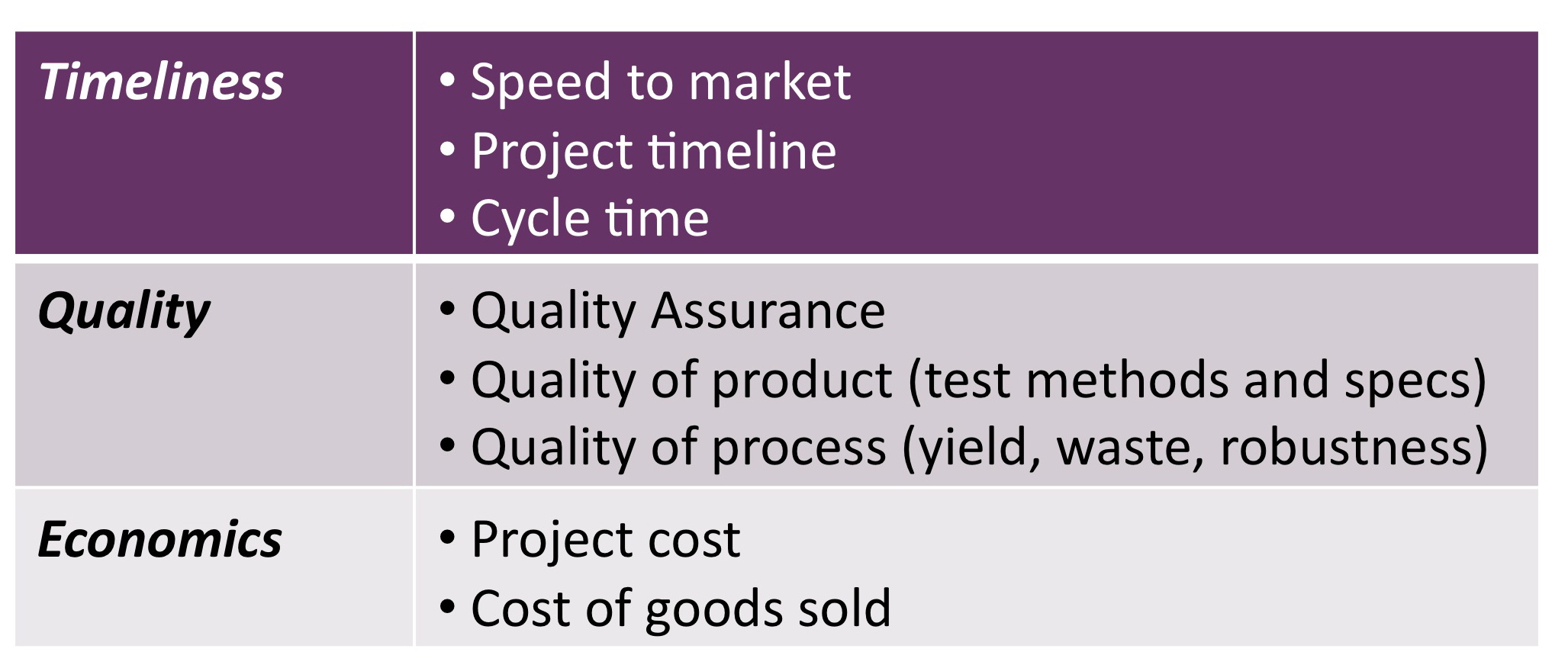
- help you strategically manage the timeliness, quality and economics of your product and programs, considering your customers (e.g., FDA, physicians, licensing partners)
- help you with your hard decisions (isn’t saying NO so much harder than saying YES?)
- be an independent set of eyes and ears for you
- work, on your behalf, as an extra set of hands to get things done
Pick-Two Consultancy has…..
- experience spanning drug discovery, chemical and pharmaceutical process development and commercial technical operations, with a strong appreciation for the non-CMC elements of development (e.g., clinical, preclinical, marketing)
- been in an entrepreneurial environment (7th employee at Sepracor) and in a fully integrated pharmaceutical company
- managed the development of a wide range of CM&C projects (oral solids, sterile solutions and metered dose inhalers), using a highly virtual model, culminating in 5 NDAs filings, 6 product launches and 2 successful out-licenses.
- a track record of creative problem solving with a broad scientific background and strong appreciation for business needs and risk management, experience at balancing internal resources and external vendors
- extensive experience with FDA (INDs, NDAs, meetings and negotiation) and drug development teams (pre-clinical, clinical, regulatory)
- built and maintained constructive, collaborative relationships with internal customers (e.g., Development teams, Marketing, Business Development, Legal, Finance) and external collaborators (e.g., FDA, CROs, CMOs, Alliance partners)
Have you discussed any of the following questions?
- How long should or can we use the discovery route?
- How pure (or impure) should our toxicology supplies be?
- Should we use a commercial-like formulation for certain clinical supplies or is a more simple formulation adequate?
- When should we “lock-in” our commercial process?
- How much process control is enough and how much is too much?
- When do we need to scale-up the process?
- How much should I invest in development a commercial process? When do we need to start?
- When should we start validation and can we sell the product?
- How much inventory (API and finished product) should we build for launch and when should we start?
- Does the benefit of the proposed post-approval change outweigh the cost?